
Article
Side Effects Reported by Jordanian Healthcare Workers Who
Received COVID-19 Vaccines
Osama Abu-Hammad
1,2
, Hamza Alduraidi
3
, Shaden Abu-Hammad
4
, Ahmed Alnazzawi
1
, Hamzah Babkair
1
,
Abdalla Abu-Hammad
5
, Ibrahim Nourwali
1
, Farah Qasem
6
and Najla Dar-Odeh
1,2,
*
Citation: Abu-Hammad, O.;
Alduraidi, H.; Abu-Hammad, S.;
Alnazzawi, A.; Babkair, H.;
Abu-Hammad, A.; Nourwali, I.;
Qasem, F.; Dar-Odeh, N. Side Effects
Reported by Jordanian Healthcare
Workers Who Received COVID-19
Vaccines. Vaccines 2021, 9, 577.
https://doi.org/10.3390/
vaccines9060577
Academic Editors:
Luis Martinez-Sobrido and
Stefano D’Errico
Received: 23 April 2021
Accepted: 22 May 2021
Published: 1 June 2021
Publisher’s Note: MDPI stays neutral
with regard to jurisdictional claims in
published maps and institutional affil-
iations.
Copyright: © 2021 by the authors.
Licensee MDPI, Basel, Switzerland.
This article is an open access article
distributed under the terms and
conditions of the Creative Commons
Attribution (CC BY) license (https://
creativecommons.org/licenses/by/
4.0/).
1
College of Dentistry, Taibah University, Al Madinah Al Munawara 43353, Saudi Arabia;
[email protected] (I.N.)
2
School of Dentistry, University of Jordan, Amman 11942, Jordan
3
School of Nursing, University of Jordan, Amman 11942, Jordan; [email protected]
4
Comprehensive Amman Healthcare Center, Amman 11192, Jordan; [email protected]
5
School of Medicine, University of Jordan, Amman 11942, Jordan; [email protected]
6
University of Jordan Hospital, Amman 11942, Jordan; [email protected]
* Correspondence: [email protected]
Abstract:
Background Distribution of COVID-19 vaccines has been surrounded by suspicions and
rumors making it necessary to provide the public with accurate reports from trustworthy experts such
as healthcare professionals. Methods We distributed a questionnaire in Jordan among physicians,
dentists and nurses who received a COVID-19 vaccine to explore the side effects (SE) they encountered
after the first or the second dose of one of three vaccines namely: AstraZeneca Vaxzevria (AZ), Pfizer-
BioNTeck (PB), and SinoPharm (SP) vaccines. Results A total of 409 professionals participated.
Approximately 18% and 31% of participants reported no SE after the first dose and second dose,
respectively. The remainder had mostly local side effects related to injection site (74%). Systemic
side effects in the form of fatigue (52%), myalgia (44%), headache (42%), and fever (35%) prevailed
mainly after the first dose. These were significantly associated with AZ vaccine, and age
≤
45 years
(p = 0.000
and 0.01, respectively). No serious SE were reported. Conclusions We can conclude that SE
of COVID-19 vaccines distributed in Jordan are within the common range known so far for these
vaccines. Further studies are needed to include larger sample size and longer follow-up period to
monitor possible serious and long-term SE of the vaccines.
Keywords:
COVID-19; AstraZeneca vaccine; Pfizer-Bionteck vaccine; SinoPharm; side effects; health-
care professionals; physicians; nurses; dentists
1. Introduction
The long-awaited vaccines of COVID-19 (corona virus disease-2019) were not received
by cheers in many areas of the world whether in developed or developing countries where
a substantial proportion of populations cast their doubts and suspicions [
1
]. Many rumors
and misinterpretations surrounded the different vaccines to variable degrees.
The devastating pandemic caused by severe acute respiratory syndrome coronavirus
2 (SARS-CoV-2) has been associated with large numbers of cases, mortalities and severe
long-term complications influencing individuals and economies.
In Jordan, the first cases of COVID-19 appeared in March 2020, but due to strict mea-
sures adopted by the government, the first wave was mild, and outcomes of the pandemic
were relatively trivial [
2
]. However, the country was hit hard by a more aggressive second
wave during September 2020. As of mid-April 2021, the 10-million population country
had over 672,000 total cases (65,364 cases per million) and 7937 deaths (772 per million) [
3
].
Jordan was among the countries that had an early start in vaccination campaigns which
started in January 2021. Taking into consideration that older adults (aged
≥
70 years) are
Vaccines 2021, 9, 577. https://doi.org/10.3390/vaccines9060577 https://www.mdpi.com/journal/vaccines

Vaccines 2021, 9, 577 2 of 10
at increased risk of severe disease and death if they develop COVID-19 [
4
], these were
prioritized for vaccination. Healthcare workers were also selected for vaccination according
to their age, hence, there were several COVID-19 related deaths registered among them.
Although many physicians were reported dead due to infection, the nation was
shocked to receive the news of a young female physician who contracted infection and
later was deceased due to the infection in late March 2021. Consequently, the Jordanian
government intensified the vaccination campaigns and initiated a campaign that specifically
targets all healthcare workers regardless their age. A nationwide campaign was established
by the Ministry of Health and under a close supervision by armed forces to provide vaccines
free of charge to all healthcare personnel.
Three types of vaccines are being administered currently in Jordan according to
availability: AstraZeneca Vaxzevria, Pfizer-BioNTeck (PB), and SinoPharm (SP) vaccines.
SE to COVID-19 vaccines were, and still are, a controversial issue and a continuously
evolving situation. According to Centers of Disease Control and Prevention (CDC), the
common adverse effects include pain, swelling and redness at injection site, as well as
fatigue, chills, fever, myalgia, headache, and nausea [5].
The vaccination campaign in Jordan was counteracted by another campaign spreading
doubts around the efficacy of these vaccines and intensifying fears around their safety
profile. Despite the large numbers of COVID-19 cases and the early start of the vaccination
campaign in Jordan, there is little data on either clinical outcomes of infection or even the
SE of vaccines. It is important at this stage to collect data on these SE of distributed vaccines
so as to inform and educate the public on this issue. More importantly, it is essential to
collect this data from trustworthy and expert sources who are willing to provide accurate
and transparent data that is not affected by geopolitical vaccine wars.
Therefore, we invited Jordanian healthcare professionals including physicians, nurses
and dentists to report the SE they encountered after receiving one or two doses of COVID-19
vaccines.
2. Materials and Methods
This was a cross-sectional survey utilizing an online questionnaire. The questionnaire
was anonymous, and it was composed of eight closed-ended questions on gender, age,
occupation, vaccine type, observed side effects and their duration after first dose and
second dose. The questionnaire was distributed among three categories of healthcare
personnel in Jordan: nurses, dentists and physicians.
The study was ethically approved by the ethical committee, School of Nursing, Uni-
versity of Jordan IRB#PF.21.14.
Statistical Analysis
IBM-SPSS statistical package for social sciences version 21 (IBM Corp: Armonk,
NY, USA) was used to obtain descriptive statistics and significant associations between
variables.
Descriptive statistics were reported as frequencies and percentages, means and stan-
dard deviations. Cross tabulation with chi square test was carried out to identify significant
associations between severity of side effects as the dependent variable and independent
variables of vaccine type, gender and age.
3. Results
The study sample consisted of 409 healthcare personnel (nurses, dentists or doctors)
working in Jordan.
3.1. Participants’ Characteristics
Age, gender distribution, profession, and vaccine types received are displayed in
Table 1.
Vaccines 2021, 9, 577 3 of 10
Table 1. Study sample sociodemographics (N = 409) and types of administered vaccines.
Variable
Dose 1 Recipients
Total (409)
Dose 2 Recipients
Total (195)
Age (years) (Mean ± SD) 34.99 ± 12.07 39.27 ± 12.79
Number (%) Number (%)
Gender
Male 120 (29.3%) 67 (34.4%)
Female 289 (70.7%) 128 (65.6%)
Profession
Physician 144 (35.2%) 71 (36.4%)
Dentist 172 (42.1%) 70 (35.9%)
Nurse 93 (22.7%) 54 (27.7%)
Vaccine type
AZ 179 (43.8%) 7 (3.6%)
PB 141 (34.5%) 101 (51.8%)
SP 89 (21.8%) 87 (44.6%)
AZ: AstraZeneca Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
3.2. Reported Side Effects
Number of side effects reported after receiving the first dose ranged between 0 and
11 for each participant, with a mean of 3.2
±
2.7 per participant. Number of side effects
reported after receiving second dose ranged between 0 and 9, with a mean of 1.4
±
2 per
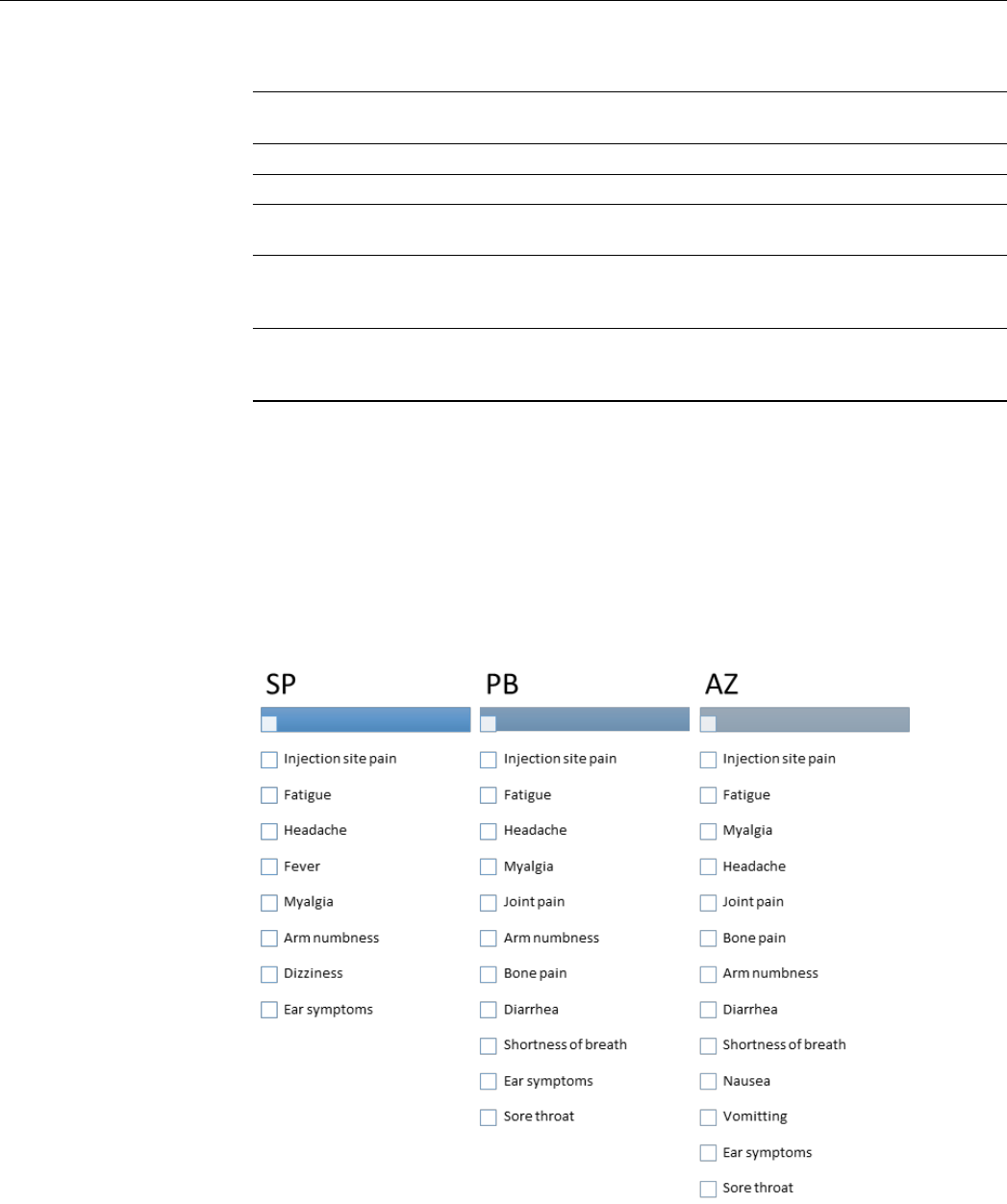
participant. Figure 1 presents the reported SE of each vaccine listed according to their
frequency and Tables 2 and 3 present frequency (%) of side effects reported after first and
second doses, and statistical significance of their association with various vaccine types
after the first dose and second dose separately.
Vaccines 2021, 9, x FOR PEER REVIEW 3 of 11
3.1. Participants’ characteristics
Age, gender distribution, profession, and vaccine types received are displayed in Ta-
ble 1.
Table 1. Study sample sociodemographics (N = 409) and types of administered vaccines.
Variable
Dose 1 Recipients
Total (409)
Dose 2 Recipients
Total (195)
Age (years) (Mean ± SD)
34.99 ± 12.07
39.27 ± 12.79
Number (%)
Number (%)
Gender
Male
120 (29.3%)
67 (34.4%)
Female
289 (70.7%)
128 (65.6%)
Profession
Physician
144 (35.2%)
71 (36.4%)
Dentist
172 (42.1%)
70 (35.9%)
Nurse
93 (22.7%)
54 (27.7%)
Vaccine type
AZ
179 (43.8%)
7 (3.6%)
PB
141 (34.5%)
101 (51.8%)
SP
89 (21.8%)
87 (44.6%)
AZ: AstraZeneca Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
3.2. Reported Side Effects
Number of side effects reported after receiving the first dose ranged between 0 and
11 for each participant, with a mean of 3.2 ± 2.7 per participant. Number of side effects
reported after receiving second dose ranged between 0 and 9, with a mean of 1.4 ± 2 per
participant. Figure 1 presents the reported SE of each vaccine listed according to their
frequency and Tables 2 and 3 present frequency (%) of side effects reported after first and
second doses, and statistical significance of their association with various vaccine types
after the first dose and second dose separately.
Figure 1. Reported side effects of vaccines arranged according to their frequency. AZ: AstraZeneca
Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
Figure 1.
Reported side effects of vaccines arranged according to their frequency. AZ: AstraZeneca
Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
Vaccines 2021, 9, 577 4 of 10
Table 2.
Side effects (number/percentage) occurring after each vaccine, and statistical significance of their association with
various vaccine types after dose 1 (number = 409).
Side Effects
Vaccine Type
Total
Number (%)
p-Value
SP
Number (%)
PB
Number (%)
AZ
Number (%)
SP-PB SP-AZ PB-AZ
Side effects
No side effects
41 (46.1%)
48 (53.9%)
118 (83.7%)
23 (16.3%)
175 (97.8%)
4 (2.2%) 75 (18.3%)
0.000 0.000 0.000
Fever
No
Yes
81 (91.0%)
8 (9.0%)
132 (93.6%)
9 (6.4%)
47 (26.3%)
132 (73.7%) 149 (36.4%)
0.462 0.000 0.000
Fatigue
No
Yes
75 (84.3%)
14 (15.7%)
96 (68.1%)
45 (31.9%)
27 (15.1%)
152 (84.9%) 211 (51.6%)
0.006 0.000 0.000
Myalgia
No
Yes
83 (93.3%)
6 (6.7%)
111 (78.7%)
30 (21.3%)
36 (20.1%)
143 (79.9%) 179 (43.8%)
0.003 0.000 0.000
Bone pain
No
Yes
89 (100.0%)
0 (0.0%)
131 (92.9%)
10 (7.1%)
100 (55.9%)
79 (44.1%) 89 (21.8%)
0.010 0.000 0.000
Joint pain
No
Yes
87 (97.8%)
2 (2.2%)
122 (86.5%)
19 (13.5%)
77 (43.0%)
102 (57.0%) 123 (30.1%)
0.004 0.000 0.000
Headache
No
Yes
78 (87.6%)
11 (12.4%)
102 (72.3%)
39 (27.7%)
56 (31.3%)
123 (68.7%) 173 (42.3%)
0.006 0.000 0.000
Injection site pain
No
Yes
57 (64.0%)
32 (36.0%)
34 (24.1%)
107 (75.9%)
16 (8.9%)
163 (91.1%) 302 (73.8%)
0.000 0.000 0.000
Arm numbness
No
Yes
86 (96.6%)
3 (3.4%)
124 (87.9%)
17 (12.1%)
138 (77.1%)
41 (22.9%) 61 (14.9%)
0.023 0.000 0.012
Diarrhea
No
Yes
89 (100.0%)
0 (0.0%)
139 (98.6%)
2 (1.4%)
162 (90.5%)
17 (9.5%) 19 (4.6%)
0.259 0.003 0.002
Shortness of breath
No
Yes
89 (100.0%)
0 (0.0%)
139 (98.6%)
2 (1.4%)
162 (90.5%)
17 (9.5%) 19 (4.6%)
0.259 0.003 0.002
Dizziness
No
Yes
88 (98.9%)
1 (1.1%)
141 (100.0%)
0 (0.0%)
173 (96.6%)
6 (3.4%) 7 (1.7%)
0.207 0.281 0.028
Vomiting
No
Yes
89 (100.0%)
0 (0.0%)
141 (100.0%)
0 (0.0%)
173 (96.6%)
6 (3.4%) 6 (1.5%)
No
vomit-
ing
0.081 0.028
Nausea
No
Yes
89 (100.0%)
0 (0.0%)
141 (100.0%)
0 (0.0%)
168 (93.9%)
11 (6.1%) 11 (2.7%)
No
nausea
0.017 0.003
Ear symptoms
No
Yes
88 (98.9%)
1 (1.1%)
140 (99.3%)
1 (0.7%)
177 (98.9%)
2 (1.1%) 4 (1%)
0.742 0.996 0.707
Sore throat
No
Yes
89 (100.0%)
0 (0.0%)
140 (99.3%)
1 (0.7%)
178 (99.4%)
1 (0.6%) 2 (0.5%)
0.426 0.480 0.865
AZ: AstraZeneca Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
Vaccines 2021, 9, 577 5 of 10
Table 3.
Side effects (number/percentage) occurring after each vaccine, and statistical significance of their association with
various vaccine types after dose 2 (number = 195).
Side Effects Dose 2
VACCINE TYPE
Total
Number (%)
p-Values
SP
Number (%)
PB
Number (%)
AZ
Number (%)
SP-PB SP-AZ PB-AZ
Side effects present
No side effects
47 (52.8%)
42 (47.2%)
125 (88.7%)
16 (11.3%)
176 (98.3%)
3 (1.7%) 61 (31.3%)
0.000 0.000 0.000
Headache
No
Yes
78 (87.6%)
11 (12.4%)
104 (73.8%)
37 (26.2%)
177 (98.9%)
2 (1.1%) 50 (25.6%)
0.012 0.000 0.000
Fever
No
Yes
85 (95.5%)
4 (4.5%)
110 (78.0%)
31 (22.0%)
176 (98.3%)
3 (1.7%) 38 (19.5%)
0.000 0.173 0.000
Fatigue
No
Yes
74 (83.1%)
15 (16.9%)
82 (58.2%)
59 (41.8%)
178 (99.4%)
1 (0.6%) 75 (38.5%)
0.000 0.000 0.000
Myalgia
No
Yes
76 (85.4%)
13 (14.6%)
104 (73.8%)
37 (26.2%)
178 (99.4%)
1 (0.6%) 51 (26.2%)
0.037 0.000 0.000
Injection site pain
no
yes
56 (62.9%)
33 (37.1%)
66 (46.8%)
75 (53.2%)
175 (97.8%)
4 (2.2%) 112 (57.4%)
0.017 0.000 0.000
Numbness injection
site
No
Yes
84 (94.4%)
5 (5.6%)
132 (93.6%)
9 (6.4%)
178 (99.4%)
1 (0.6%) 15 (7.7%)
0.813 0.008 0.003
Joint pain
No
Yes
86 (96.6%)
3 (3.4%)
118 (83.7%)
23 (16.3%)
178 (99.4%)
1 (0.6%) 27 (13.8%)
0.003 0.074 0.000
Diarrhea
No
Yes
88 (98.9%)
1 (1.1%)
137 (97.2%)
4 (2.8%)
178 (99.4%)
1 (0.6%) 6 (3.1%)
0.386 0.613 0.103
Shortness of breath
No
Yes
87 (97.8%)
2 (2.2%)
133 (94.3%)
8 (5.7%)
179 (100.0%)
0 (0.0%) 10 (5.1%)
0.215 0.044 0.001
Bone pain
No
Yes
85 (95.5%)
4 (4.5%)
122 (86.5%)
19 (13.5%)
177 (98.9%)
2 (1.1%) 25 (12.8%)
0.027 0.078 0.000
Three participants had COVID-19 infection after the first dose of PB vaccine and two had the infection after the AZ vaccine, however, the p
value was insignificant (p = 0.354). Additionally, two had the infection after the second dose of the SP vaccine and one after the PB vaccine,
however, the p value was not significant (p = 0.127). AZ: AstraZeneca Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
Table 4 presents less frequent side effects that occurred after first or second dose for
all vaccines.
3.3. Relationship of Severity of Side Effects with Age and Gender and Vaccine Type
Severity was expressed as no reported side effects, local side effects (injection site pain
or arm numbness) and systemic side effects (all remaining side effects). Cross tabulation
of severity of side effects with type of vaccine following both doses, showed that AZ was
found to be significantly associated with more severe side effects, while SP was significantly
associated with no side effects and PB was significantly associated with local side effects
(p = 0.000) (Table 5). Age group but not gender was significantly associated with severity
of SE after first dose only (p = 0.01), however, no statistically significant association was
found between either gender or age group and severity of SE after second dose (p > 0.05).
Vaccines 2021, 9, 577 6 of 10
Table 4. Frequency (%) of less frequent side effects after dose 1 and dose 2 of various vaccines.
Side Effect
Vaccine Type
Total
N = 409
SP (N = 89)
N (%)
PB (N = 141)
N (%)
AZ (N = 179)
N (%)
First dose
Chest pain 0 0 1 (0.6%) 1 (0.2%)
Common cold 1 (1.1%) 0 0 1 (0.2%)
Cough 0 0 1 (0.6%) 1 (0.2%)
Herpes zoster 0 1 (0.7%) 0 1 (0.2%)
Loss of smell 0 1 (0.7%) 0 1 (0.2%)
Lower back pain 0 0 1 (0.6%) 1 (0.2%)
Palpitations 0 0 1 (0.6%) 1 (0.2%)
Redness and swelling (injection site) 0 1 (0.7%) 0 1 (0.2%)
Sleepiness 1 (1.1%) 0 0 1 (0.2%)
Thirst 0 0 1 (0.6%) 1 (0.2%)
Urticaria 0 0 1 (0.6%) 1 (0.2%)
Burning pain in head 0 1 (0.7%) 0 1 (0.2%)
Loin pain 0 0 1 1 (0.2%)
Second dose
Cough 1 (1.1%) 0 0 1 (0.2%)
Diuresis 0 2 (1.4%) 0 2 (0.5)
Herpes zoster 1 (1.1%) 0 0 1 (0.2%)
Sleepiness 1 (1.1%) 0 0 1 (0.2%)
Table 5. Cross tabulation of severity of side effects after doses 1 and 2 with age groups, gender and vaccine type.
Severity of Side Effects after Dose 1 No Side Effects Local Systemic p-Value
Age (years)
≤45
>45
45 (15.8%)
2 (30.3%)
70 (24.6%)
19 (21.3%)
170 (59.6%)
43 (48.3%) 0.010
Gender
Male
Female
23 (22.1%)
49 (18.1%)
3 (28.8%)
59 (21.9%)
51 (49.0%)
162 (60.0%)
0.154
Vaccine type
SP
PB
AZ
48 (60.0%)
21 (16.4%)
3 (1.8%)
20 (25.0%)
59 (46.1%)
10 (6.0%)
12 (15.0%)
48 (37.5%)
153 (92.2%)
0.000
Severity of side effects after dose 2
Age (years)
≤45
>45
37 (28.9%)
24 (35.8%)
20 (15.6%)
14 (20.9%)
71 (55.5%)
29 (43.3%)
0.266
Gender
Male
Female
22 (32.8%)
39 (30.5%)
8 (11.9%)
26 (20.3%)
37 (55.2%)
63 (49.2%)
0.340
Vaccine type
SP
PB
AZ
42 (48.3%)
16 (15.8%)
3 (42.9%)
19 (21.8%)
15 (14.9%)
0 (0.0%)
26 (29.9%)
70 (69.3%)
4 (57.1%) 0.000
AZ: AstraZeneca Vaxezevira; PB: Pfizer BioNTech vaccine, SP: SinoPharm vaccine.
3.4. Duration of Systemic Side Effects after Dose 1 in Days
On average systemic side effects lasted for 1.39
±
1.12 days, with a range of 0–5 days.
When the patient reported recovery from severe SE within 24 h of their start, the duration
was calculated as 0 days.
4. Discussion
We conducted this study to explore the side effects of COVID-19 vaccines among their
recipients of healthcare professionals to ensure, as much as possible, the accuracy and
credibility of data collected. Physicians, nurses, and dentists were invited to participate

Vaccines 2021, 9, 577 7 of 10
in this survey because we think that these professionals are strongly related to the critical
health aspects of the pandemic. Physicians and nurses work in the front lines in the combat
against the pandemic, whereas dentists are at a high risk to contract the infection due to
the intimate and risky nature of dental practice in dealing with highly infectious fluids and
aerosols [6].
Until now vaccination campaigns against COVID-19 have been influenced by rumors,
suspicions, hesitancy and refusal. There was also exaggeration and over-reporting of
adverse effects of vaccines, as some of these effects are normal physiologic processes or
developmental anomalies that cannot be related in any way to any drug and definitely not
to vaccines. Some reported SE are either normal physiologic processes such as teething,
while others could be developmental conditions such as fissured tongue [
7
]. Monitoring
the safety of COVID-19 vaccines is an important and ongoing process that should also
be accurate. In the US, Vaccine Adverse Event Reporting System has been implemented
as an active surveillance system, during the initial implementation phases of the COVID-
19 national vaccination program [
8
]. A similar system is being adopted in Europe by
individual national authorities, in collaboration with the European Centre for Disease
Prevention and Control and European Medicine Agency (EMA) [9].
In Jordan at least 524,533 doses of COVID-19 vaccines have been administered as of
15 April 2021, which are enough to vaccinate about 2.6% of the population utilizing the
two-dose regimen [
10
]. Unfortunately, little research has been published on COVID-19
clinical outcomes in Jordan and as far as we know no research has been conducted there on
either efficacy or safety of its vaccines so far.
The study included three vaccines and it investigated SE after first and second doses
separately because we aimed to collect as many responses as possible, and to explore
possible variations in reactions between first and second doses. It is important at this critical
stage of the vaccination campaign to reassure vaccine recipients by collecting evidence-
based data about the local and systemic SE especially if these effects have a transient or
temporary nature which might abolish fears and encourage completion of the two-dose
vaccination series [
8
], and booster doses in the future if need arises. The three vaccines
included in this study were PB (an mRNA vaccine), AZ vaccine-Vaxzevria, (adenovirus
vector encoding the S glycoprotein of SARS-CoV-2), and SP (inactivated vaccine). PB and
AZ vaccines are being distributed on a wide scale and their SE are being continuously
documented and reported. On the other hand, there is little information on SP vaccine in
terms of its efficacy and safety. Currently, a limited number of countries adopted SP in
their vaccination campaigns including Jordan, United Arab Emirates, Bahrain, Egypt and
Peru [11]. Hence, it is important to provide data on this particular vaccine as well.
Approximately one in five and one in three did not have side effects after the first
and second dose, respectively. The lower rate of side effects in second dose recipients
noticed in this study is probably correlated to the variation in numbers of various vaccine
recipients among first and second dose recipients. Less than 4% of the second dose
recipients (compared to 44% of first dose recipients) have received AZ vaccine which was
found to be the one highly associated with side effects. On the other hand, approximately
45% of second dose recipients have received SP vaccine, the one with minimal side effects.
Most participants who received SP vaccine did not report any side effects after any of
the two doses indicating the weak immunogenic potential of the vaccine. Inactivated
vaccines are said to have a good safety profile, however, they need a booster plan to create
immune memory [
12
]. Most participants had local side effects associated with post-injection
pain and numbness. Among systemic side effects, fatigue represented the most common
symptom after both first and second doses. Other cited side effects included myalgia,
headache, fever, arthralgia, and bone pain. According to Centers of Disease Control
and Prevention (CDC), the common adverse effects of COVID-19 vaccines include pain,
swelling and redness at injection site, as well as fatigue, chills, fever, myalgia, headache,
and nausea [
5
]. A minority of participants reported gastrointestinal side effects (nausea,

Vaccines 2021, 9, 577 8 of 10
vomiting, diarrhea), respiratory side effects (dyspnea), and there were some sporadic cases
of ear symptoms, facial pain, sleepiness, and diuresis.
PB vaccine SE were reported in literature as fatigue, chills, headache, myalgia, and
pain at the injection site. Such adverse events were dose-dependent and were more
common after the second immunization [
13
]. This is consistent with our study wherein
PB vaccine was significantly associated with headache, fever, fatigue, myalgia and joint
pain after the second dose. This vaccine can also rarely cause anaphylactic shock [
14
].
According to Medicines and Healthcare Products Regulatory Agency (MHRA), it has been
advised to administer the vaccine with caution to individuals who have any history of an
allergic reaction to a vaccine, drug or food and especially people who need an adrenaline
auto-injector in emergency cases [
15
]. In comparison to other COVID-19 vaccines it is more
temperature sensitive, consequently, it is harder to store and transport [
14
]. None of our
participants reported allergic reaction to PB vaccine.
The most severe side effects were correlated with AZ vaccine (>90% of vaccine recipi-
ents), while the PB was significantly associated with severity of local side effects (46% of
PB vaccine recipients). It was reported that Vaxzevria had mild adverse reactions including
chills, fatigue, headache, fever, nausea, muscle aches, malaise, and painful injection sites
within a week post-vaccination [
15
]. As a prophylactic measure, paracetamol was recom-
mended to reduce these post-vaccination local and systemic reactions [16]. It was noticed
that there were not significant differences between males and females in development of
systemic side effects, however, participants who were 45 years or younger had significantly
more systemic side effects. Vaxzevria was described as being better tolerated in older
adults than in younger adults and has similar immunogenicity across all age groups after
a boost dose [
4
]. Most of the reported local and systemic adverse events in the literature
were mild to moderate in severity, and it was found that booster vaccination was associated
with fewer adverse effects than the first dose of vaccination and reactogenicity reduced
with increasing age [
4
]. Age restrictions in vaccine selection have been applied in several
countries. Although the MHRA recommended against age restrictions in AZ vaccine use,
the Joint Committee on Vaccination and Immunisation (JCVI) in the UK advised that people
younger than 30 should be offered alternative vaccines where available [
17
]. Currently,
several countries inside and outside Europe have put age restrictions to AZ vaccine recip-
ients [
17
]. If there are going to be age restrictions in Jordan as well, there is a possibility
that a large sector of the population will be offered other alternatives because the median
age is estimated to be 23.8 years. Of interest are the gastrointestinal symptoms affecting
recipients of AZ vaccine. The vaccine was found to be clearly associated with diarrhea,
nausea and vomiting. This may be explained by the nature of this vaccine (S glycoprotein)
and its influence on the gastrointestinal system, an influence that was previously elucidated
for SARS-CoV-2 itself [
18
]. No serious SE are being reported by our study. According
to EMA, 30 cases of thromboembolic events (predominantly venous) had been reported
by 10 March 2021, among the approximately 5 million recipients of the Vaxzevria in the
European Economic Area [
19
]. It is recommended that people who have persistent and
very severe headaches within the first 2–3 weeks after vaccination get further evaluation.
A safety update released by the EMA on 16 April 2021 specified thrombocytopenia as a
new common side effect (in <1 in 10 persons) and thrombosis in combination with throm-
bocytopenia as a new very rare side effect (in <1 in 10,000 persons). Side effects included
shortness of breath, chest or persistent abdominal pain, leg swelling, severe or persistent
headache, blurred vision, persistent bleeding, and skin bruising or round, pinpoint spots
beyond the site of vaccination appearing after a few days [
20
]. Recently, Østergaard et al.
(2021) did a nationwide analysis of population-based data from Denmark to estimate the
natural incidence of venous thromboembolism and compare it to that related to Vaxzevria,
and there was no difference [
21
]. In tackling this issue, it is important to address the
difference in sociodemographics and lifestyle factors among different nations. In Jordan
other possible confounders may exist. The role of several psychological triggers related to
the COVID-19 pandemic was suggested in a recent study investigating the development of

Vaccines 2021, 9, 577 9 of 10
acute myocardial infarction in Jordanian non-COVID-19 patients [
22
]. These confounders
were found to be lockdown stress, loneliness, unstable income, unemployment, binge
smoking, anger and fear of contracting COVID-19 infection [
22
]. Some participants in this
study reported headache, dyspnea, tinnitus and facial pain. Whether this is related to the
vaccine needs to be further verified. Two cases of herpes zoster were also reported as a side
effect in this study. This infection, as well as several recurrent herpes infections, is a known
complication for depressed immunity. The influence of the SARS-CoV-2 on the immune
system, and its ability to stimulate recurrent opportunistic viral infections of the skin and
mucous membranes are already established [
23
]. Similarly, it seems that the influence of
the vaccine on the immune system should not be underestimated.
Our study included three vaccines, two of them have attracted attention worldwide.
SP vaccine has been under the spotlight because of its rather “quiet” profile, while AZ
vaccine has been surrounded by noise in several pivotal areas of the world. The results of
this study confirmed that SP is really a “quiet” vaccine since it was significantly associated
with symptom-free vaccination. However, these findings neither confirm its efficacy, nor
exclude its long-term SE. On the other hand, AZ vaccine was found to be most significantly
associated vaccine with post vaccination side effects. However, it should be noted that no
serious SE were reported, and that recovery in all affected participants was achieved in a
duration of days.
It was not the aim of this study to investigate efficacy of vaccines, however, five par-
ticipants considered COVID-19 infection as a side effect. Contracting COVID-19 infection
after being vaccinated is consistent with the less than 100% efficacy rate that the produc-
ers of COVID-19 vaccines reported after both doses. Vaccine effectiveness in preventing
COVID-19 was reported to be 95% for PB [
24
] and 70.4% for AZ vaccine [
25
]. As for SP the
picture is not yet clear, with efficacy ranging from 50% to 78% according to trials conducted
on this vaccine in some countries [11].
There were limitations in this study owing to the cross-sectional, self-reported design
and the possibility that psychological factors may have influenced participants’ perceptions.
Another limitation is that the study did not investigate medical status of participants, and
how this could have influenced their responses to the vaccines. Therefore, we cannot rule
out the influence of medical status as an important confounder in determining vaccine SE.
Not all our participants have received the second dose yet because it is to be taken in the
near future. It was important to include all those who received one or two doses due to
the urgent and ongoing nature of vaccine evaluation. Further, response rate could not be
determined because at the time of conducting this study the total number of vaccinated
healthcare workers in Jordan was not yet known. Therefore, we included a convenient
sample. The main strength of this study is that the study sample was derived from
healthcare professionals who are expected to provide transparent information based on
their medical and scientific background.
It is recommended that longitudinal surveys are conducted over the coming years
to investigate possible long-term SE of vaccines. Alongside these surveys, other surveys
should be conducted to examine the efficacy of these vaccines in preventing COVID-19,
and the best regimen in booster vaccination.
Author Contributions:
Conceptualization, O.A.-H., S.A.-H., N.D.-O.; methodology, A.A. and H.B.;
resources, A.A.-H. and F.Q.; data curation, H.A., and I.N.; writing—original draft preparation,
O.A.-H.
and A.A.-H.; writing—review and editing, N.D.-O. and S.A.-H.; ethical approval, H.A. All
authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement:
This study received unconditional approval by Research
Ethics committee of School of Nursing, University of Jordan, Amman, Jordan. IRB#PF.21.14.
Informed Consent Statement:
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement: Data can be obtained from corresponding author upon request.

Vaccines 2021, 9, 577 10 of 10
Conflicts of Interest: The authors declare no conflict of interest.
References
1.
Khan, Y.H.; Mallhi, T.H.; Alotaibi, N.H.; Alzarea, A.I.; Alanazi, A.S.; Tanveer, N.; Hashmi, F.K. Threat of COVID-19 Vaccine
Hesitancy in Pakistan: The Need for Measures to Neutralize Misleading Narratives. Am. J. Trop. Med. Hyg.
2020
, 103, 603–604.
[CrossRef] [PubMed]
2.
Abu-Hammad, O.; Alnazzawi, A.; Borzangy, S.S.; Abu-Hammad, A.; Fayad, M.; Saadaledin, S.; Abu-Hammad, S.; Dar-Odeh, N.
Factors Influencing Global Variations in COVID-19 Cases and Fatalities; A Review. Healthcare
2020
, 8, 216. [CrossRef] [PubMed]
3. WM. Available online: https://www.worldometers.info/coronavirus/#countries (accessed on 24 March 2021).
4.
Ramasamy, M.N.; Minassian, A.M.; Ewer, K.J.; Flaxman, A.L.; Folegatti, P.M.; Owens, D.R.; Voysey, M.; Aley, P.K.; Angus, B.;
Babbage, G.; et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young
and old adults (COV002): A single-blind, randomised, controlled, phase 2/3 trial. Lancet 2021, 396, 1979–1993. [CrossRef]
5.
CDC. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/expect/after.html (accessed on 17 April 2021).
6.
Dar-Odeh, N.; Babkair, H.; Abu-Hammad, S.; Borzangy, S.; Abu-Hammad, A.; Abu-Hammad, O. COVID-19: Present and Future
Challenges for Dental Practice. Int. J. Environ. Res. Public Health 2020, 17, 3151. [CrossRef] [PubMed]
7.
MHRA. Available online: https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions
(accessed on 17 April 2021).
8.
Gee, J.; Marquez, P.; Su, J.; Calvert, G.; Liu, R.; Myers, T.; Nair, N.; Martin, S.; ClarK, T.; Markowitz, L.; et al. First Month of
COVID-19 Vaccine Safety Monitoring—United States, December 14, 2020-January 13, 2021. MMWR Morb. Mortal. Wkly. Rep.
2021, 70, 283–288. [CrossRef] [PubMed]
9.
Cavaleri, M.; Enzmann, H.; Straus, S.; Cooke, E. The European Medicines Agency’s EU conditional marketing authorisations for
COVID-19 vaccines. Lancet 2021, 397, 355–357. [CrossRef]
10.
Rheuters. Available online: https://graphics.reuters.com/world-coronavirus-tracker-and-maps/countries-and-territories/
jordan/ (accessed on 15 April 2021).
11. Baraniuk, C. What do we know about China’s covid-19 vaccines? BMJ 2021, 373, n912. [CrossRef] [PubMed]
12.
Rodriguez-Coira, J.; Sokolowska, M. SARS-CoV-2 candidate vaccines—Composition, mechanisms of action and stages of clinical
development. Allergy 2020. [CrossRef]
13.
Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens,
L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med.
2020
,
383, 2427–2438. [CrossRef] [PubMed]
14.
Meo, S.A.; Bukhari, I.A.; Akram, J.; Meo, A.S.; Klonoff, D.C. COVID-19 vaccines: Comparison of biological, pharmacological
characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur. Rev. Med. Pharmacol. Sci.
2021
, 25, 1663–1669.
[PubMed]
15.
Ashraf, M.U.; Kim, Y.; Kumar, S.; Seo, D.; Ashraf, M.; Bae, Y.-S. COVID-19 Vaccines (Revisited) and Oral-Mucosal Vector System
as a Potential Vaccine Platform. Vaccines 2021, 9, 171. [CrossRef] [PubMed]
16.
Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck,
E.A. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2,
single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [CrossRef]
17. Wise, J. Covid-19: How AstraZeneca lost the vaccine PR war. BMJ 2021, 373, n921. [CrossRef] [PubMed]
18.
Elsayed, S.A.; Abu-Hammad, O.; Alolayan, A.B.; Eldeen, Y.S.; Dar-Odeh, N. Fallacies and Facts around COVID-19: The
Multifaceted Infection. J. Craniofac. Surg. 2020, 31, e643–e644. [CrossRef] [PubMed]
19.
Wise, J. Covid-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ
2021
,
372, n699. [CrossRef] [PubMed]
20.
EMA. Available online: https://www.ema.europa.eu/en/documents/covid-19-vaccine-safety-update/covid-19-vaccine-safety-
update-vaxzevria-previously-covid-19-vaccine-astrazeneca-14-april-2021_en.pdf (accessed on 16 April 2021).
21.
Ostergaard, S.D.; Schmidt, M.; Horváth-Puhó, E.; Thomsen, R.W.; Sørensen, H.T. Thromboembolism and the Oxford-AstraZeneca
COVID-19 vaccine: Side-effect or coincidence? Lancet 2021, 397, 1441–1443. [CrossRef]
22.
Hammoudeh, A.; Tabbalat, A.-H.H.; Al-Mousa, R.; Madanat, E.E.; Al-Muhaisen, R.; Fakhri, F.; Alhaddad, I. The Covid-19
Pandemic and Triggered Acute Myocardial Infarction among Non-Infected Individuals. Int. J. Clin. Cardiol.
2020
, 7, 185.
[CrossRef]
23.
Abu-Hammad, S.; Dar-Odeh, N.; Abu-Hammad, O. SARS-CoV-2 and oral ulcers: A causative agent or a predisposing factor?
Oral Dis. 2020. [CrossRef] [PubMed]
24.
Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.;
et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med.
2020
, 383, 2603–2615. [CrossRef] [PubMed]
25.
Voysey, M.; Clemens, S.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat,
Q.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four
randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [CrossRef]
